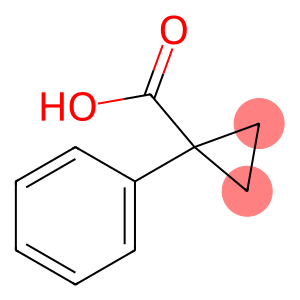
1-phenylcyclopropanecarboxylic acid
1-Phenyl-1-cyclopropanecarboxylic acid
CAS: 6120-95-2
Molecular Formula: C10H10O2
1-phenylcyclopropanecarboxylic acid - Names and Identifiers
| Name | 1-Phenyl-1-cyclopropanecarboxylic acid |
| Synonyms | AKOS BB-9362 RARECHEM AL BO 1075 LABOTEST-BB LT00451798 1-phenylcyclopropanecarboxylate 1-Phenylcyclopropanecarboxylic acid 1-phenylcyclopropanecarboxylic acid 1-PHENYL-1-CYCLOPROPANECARBOXYLIC ACID Cyclopropanecarboxylic acid, 1-phenyl- 1-Phenyl-1-cyclopropanecarboxylic acid |
| CAS | 6120-95-2 |
| EINECS | 228-090-9 |
| InChI | InChI=1/C10H10O2/c11-9(12)10(6-7-10)8-4-2-1-3-5-8/h1-5H,6-7H2,(H,11,12)/p-1 |
| InChIKey | IWWCCNVRNHTGLV-UHFFFAOYSA-N |
1-phenylcyclopropanecarboxylic acid - Physico-chemical Properties
| Molecular Formula | C10H10O2 |
| Molar Mass | 162.19 |
| Density | 1.0613 (rough estimate) |
| Melting Point | 85-87 °C (lit.) |
| Boling Point | 248.88°C (rough estimate) |
| Flash Point | 141.2°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.000277mmHg at 25°C |
| Appearance | Crystals, Crystalline Powder or Chunks |
| Color | White to beige |
| pKa | 4.30±0.20(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.5782 (estimate) |
| MDL | MFCD00001288 |
| Use | It was used in the preparation of bis[(1-phenyl-1-cyclopropyl)carbonyl] peroxide via reaction with hydrogen peroxide in the presence of N,N′-dicyclohexylcarbodiimide. |
1-phenylcyclopropanecarboxylic acid - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29163990 |
| Hazard Class | IRRITANT |
1-phenylcyclopropanecarboxylic acid - Reference Information
| Introduction | , pesticides and petrochemical industry, can be used as intermediates and other compounds, to synthesize many of the fuel, liquid crystal materials, pharmaceutical and other compounds have an important role. For example, the acidic substances synthesized with phenylpropanoic acid as the parent substance can effectively promote the spontaneous dissolution of blood fiber protease, and quickly inhibit serum poisoning; P-methoxyphenylalanine and its sodium salt, potassium salt, acetanilide and ammonium salts are a class of highly effective agricultural insecticides. Therefore, the synthesis of phenylpropanoic acid compounds has attracted much attention.|
| Application | 1-phenyl-1-cyclopropanecarboxylic acid is a derivative of phenylalanine. Phenylpropanoic acid and its derivatives are commonly found in the pharmaceutical, pesticide and petrochemical industries, and the carboxyl group of phenylpropanoic acid itself is an active group, can be used as an intermediate and other compounds reaction, so as to synthesize many in the fuel, liquid crystal materials, pharmaceutical and other aspects have an important role in the compound. |
| preparation | to the substituted phenylacetonitrile was added benzyltriethylammonium chloride (0.025 equivalents) and the appropriate dihalogenated compound (2.5 equivalents). The mixture was heated at 70 °c and then 50% sodium hydroxide (10 eq.) was slowly added to the mixture. The reaction was stirred at 70 °c for 12-24 hours to ensure complete formation of the cycloalkyl moiety and then heated at 130 °c for 24-48 hours to ensure complete conversion of the nitrile to the carboxylic acid. The dark brown/black reaction mixture was diluted with water and extracted three times each with ethyl acetate and then with dichloromethane to remove by-products. The aqueous alkaline solution was acidified with concentrated hydrochloric acid to a pH of less than one, and the precipitate starting at pH 4 was filtered and washed twice with 1M hydrochloric acid. The solid product was dissolved in dichloromethane and extracted twice with 1M hydrochloric acid and once with saturated aqueous sodium chloride solution. The organic solution was dried over sodium sulfate and evaporated to dryness to give the cycloalkyl carboxylic acid. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 1-Phenyl-1-cyclopropanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 6120-95-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Phenylcyclobutylamine Request for quotation
CAS: 6120-95-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6120-95-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 1-Phenyl-1-cyclopropanecarboxylic acid Request for quotation
CAS: 6120-95-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6120-95-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 1-Phenyl-1-cyclopropanecarboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 6120-95-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6120-95-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-Phenyl-1-cyclopropanecarboxylic acid Visit Supplier Webpage Request for quotationCAS: 6120-95-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Phenylcyclobutylamine Request for quotation
CAS: 6120-95-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6120-95-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 1-Phenyl-1-cyclopropanecarboxylic acid Request for quotation
CAS: 6120-95-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6120-95-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 1-Phenyl-1-cyclopropanecarboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 6120-95-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6120-95-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



